The smart way to manage the impact of changes in your medical devices
The Essenvia RIM Platform allows you to easily manage change assessment and streamline collaboration between regulatory and other departments (e.g. QA,R&D).
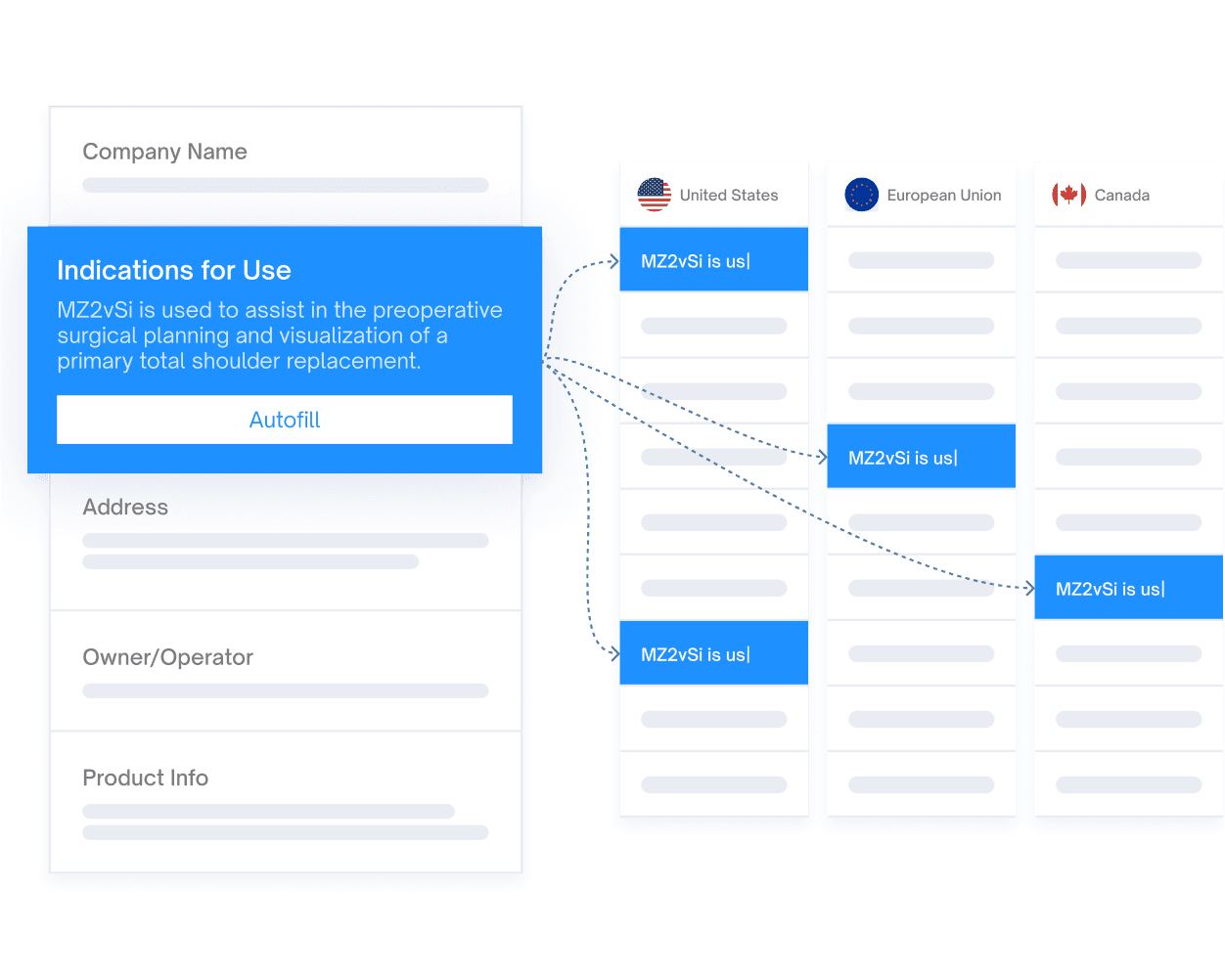
The Essenvia RIM Platform allows you to easily manage change assessment and streamline collaboration between regulatory and other departments (e.g. QA,R&D).
